QQuestionAnatomy and Physiology
QuestionAnatomy and Physiology
Potassium (K): $1 s^{2} 2 s^{2} 2 p^{6} 3 s^{2} 3 p^{6} 4 s^{1}$
Identify the core electrons and valence electrons.
Attachments
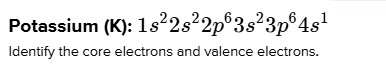
6 months agoReport content
Answer
Full Solution Locked
Sign in to view the complete step-by-step solution and unlock all study resources.
Step 1I'll solve this problem step by step, carefully following the LaTeX formatting guidelines:
Step 2: Understand the Electron Configuration
The electron configuration for Potassium (K) is $$1s^{2} 2s^{2} 2p^{6} 3s^{2} 3p^{6} 4s^{1}
Final Answer
- Core Electrons: 18 - Valence Electrons: 1
Need Help with Homework?
Stuck on a difficult problem? We've got you covered:
- Post your question or upload an image
- Get instant step-by-step solutions
- Learn from our AI and community of students