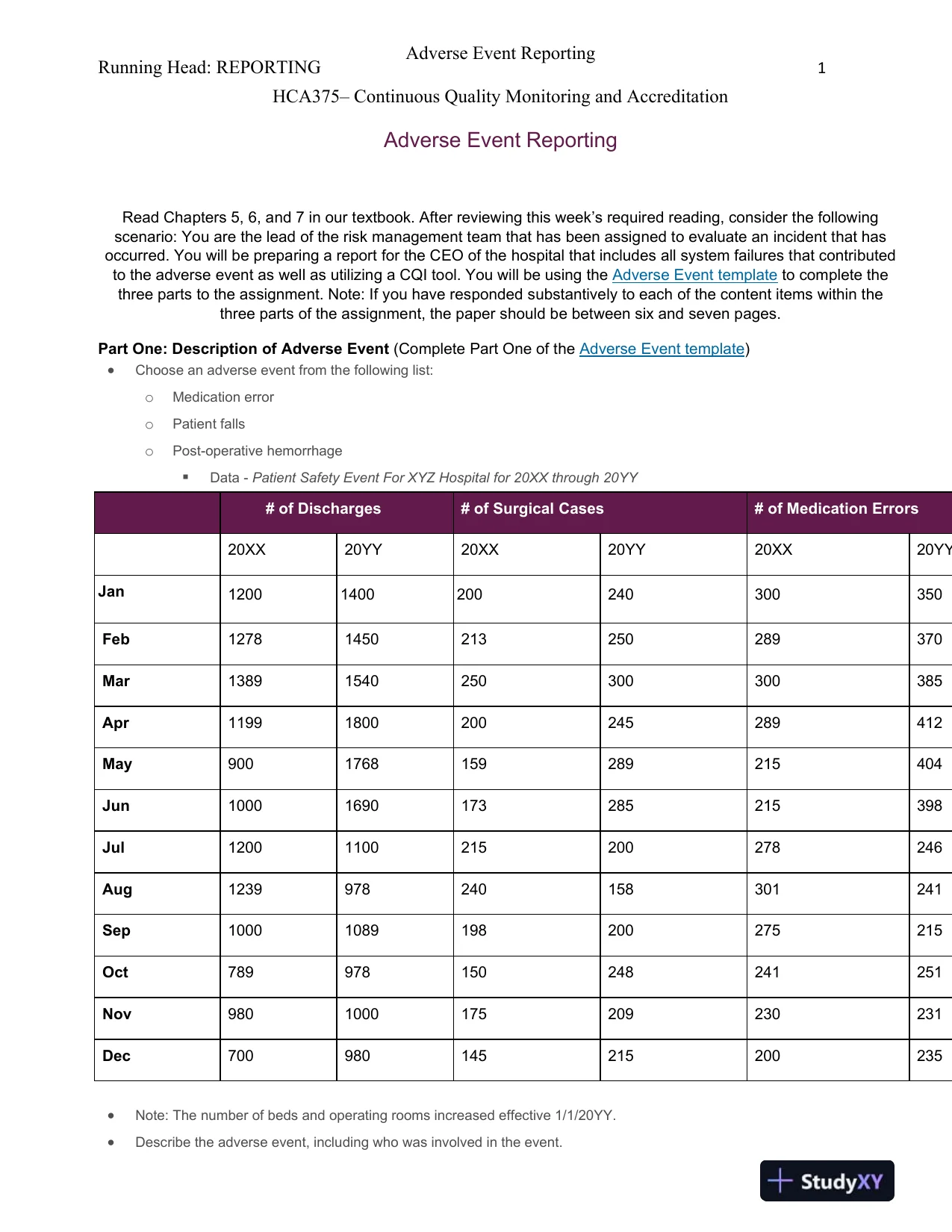
Running Head: REPORTING1Adverse Event ReportingHCA375–Continuous Quality Monitoring and AccreditationAdverse Event ReportingRead Chapters 5, 6, and 7 in our textbook. After reviewing this week’s required reading, consider the followingscenario: You arethe lead of the risk management team that has been assigned to evaluate an incident that hasoccurred. You will be preparing a report for the CEO of the hospital that includes all system failures that contributedto the adverse event as well as utilizing a CQI tool. You will be using theAdverse Event templateto complete thethree parts to the assignment. Note: If you have responded substantively to each of the content items within thethree parts of the assignment, the paper should be between six and seven pages.Part One: Description of Adverse Event(Complete Part One of theAdverse Event template)Choose an adverse event from the following list:oMedication erroroPatient fallsoPost-operative hemorrhageData-Patient Safety Event For XYZ Hospital for 20XX through 20YY# of Discharges# of Surgical Cases# of Medication Errors20XX20YY20XX20YY20XX20YYJan12001400200240300350Feb12781450213250289370Mar13891540250300300385Apr11991800200245289412May9001768159289215404Jun10001690173285215398Jul12001100215200278246Aug1239978240158301241Sep10001089198200275215Oct789978150248241251Nov9801000175209230231Dec700980145215200235Note: Thenumber of beds and operating rooms increased effective 1/1/20YY.Describe the adverse event, including who was involved in the event.
Running Head: REPORTING1Adverse Event ReportingHCA375–Continuous Quality Monitoring and AccreditationAdverse Event ReportingRead Chapters 5, 6, and 7 in our textbook. After reviewing this week’s required reading, consider the followingscenario: You arethe lead of the risk management team that has been assigned to evaluate an incident that hasoccurred. You will be preparing a report for the CEO of the hospital that includes all system failures that contributedto the adverse event as well as utilizing a CQI tool. You will be using theAdverse Event templateto complete thethree parts to the assignment. Note: If you have responded substantively to each of the content items within thethree parts of the assignment, the paper should be between six and seven pages.Part One: Description of Adverse Event(Complete Part One of theAdverse Event template)Choose an adverse event from the following list:oMedication erroroPatient fallsoPost-operative hemorrhageData-Patient Safety Event For XYZ Hospital for 20XX through 20YY# of Discharges# of Surgical Cases# of Medication Errors20XX20YY20XX20YY20XX20YYJan12001400200240300350Feb12781450213250289370Mar13891540250300300385Apr11991800200245289412May9001768159289215404Jun10001690173285215398Jul12001100215200278246Aug1239978240158301241Sep10001089198200275215Oct789978150248241251Nov9801000175209230231Dec700980145215200235Note: Thenumber of beds and operating rooms increased effective 1/1/20YY.Describe the adverse event, including who was involved in the event.Preview Mode
This document has 11 pages. Sign in to access the full document!