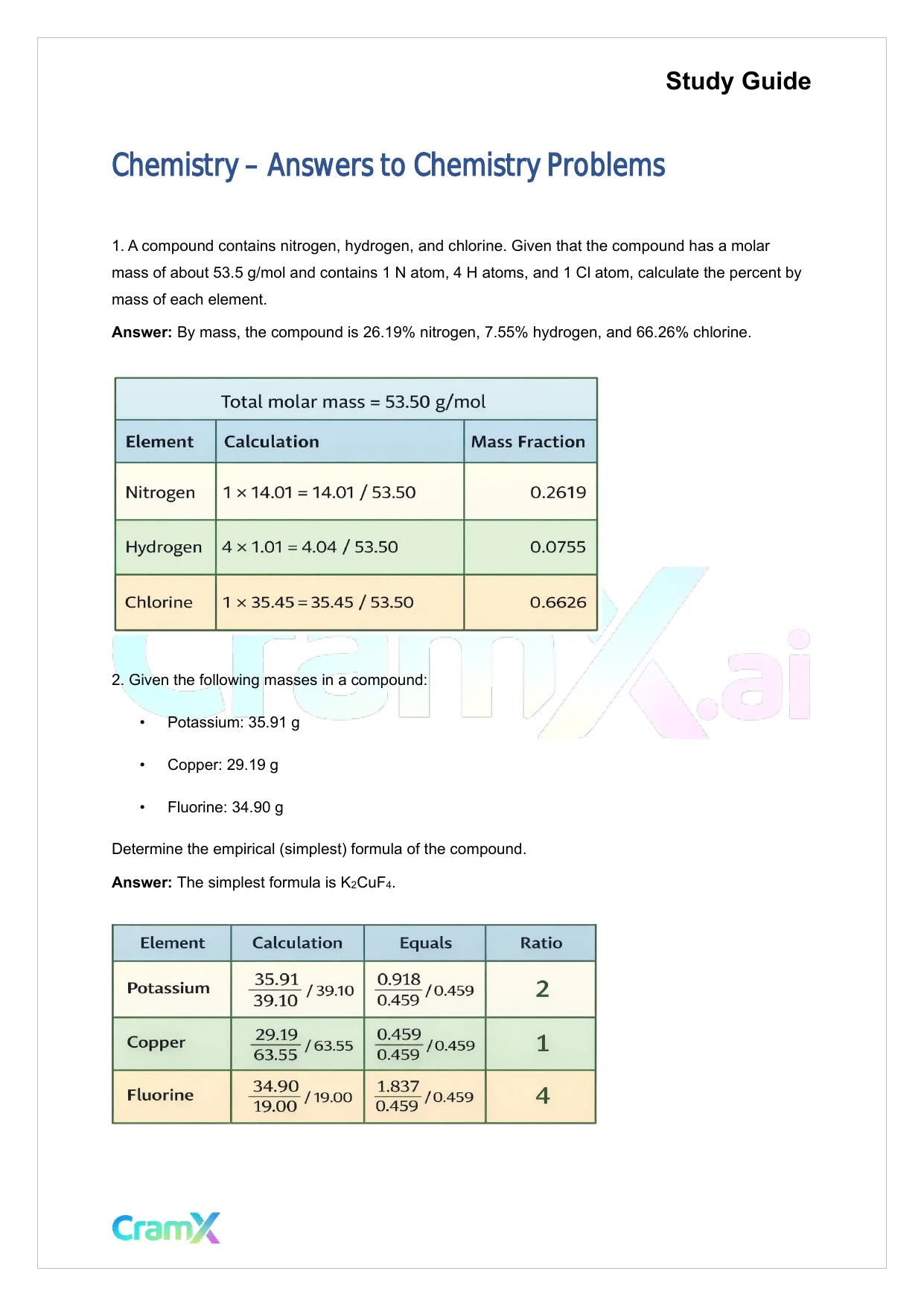
Study GuideChemistry – Answers to Chemistry Problems1.A compound contains nitrogen, hydrogen, and chlorine. Given that the compound has a molarmass of about 53.5 g/mol and contains 1 N atom, 4 H atoms, and 1 Cl atom, calculate the percent bymass of each element.Answer:By mass, the compound is 26.19% nitrogen, 7.55% hydrogen, and 66.26% chlorine.2.Given the following masses in a compound:•Potassium: 35.91 g•Copper: 29.19 g•Fluorine: 34.90 gDetermine the empirical (simplest) formula of the compound.Answer:The simplest formula is K2CuF4.
Study GuideChemistry – Answers to Chemistry Problems1.A compound contains nitrogen, hydrogen, and chlorine. Given that the compound has a molarmass of about 53.5 g/mol and contains 1 N atom, 4 H atoms, and 1 Cl atom, calculate the percent bymass of each element.Answer:By mass, the compound is 26.19% nitrogen, 7.55% hydrogen, and 66.26% chlorine.2.Given the following masses in a compound:•Potassium: 35.91 g•Copper: 29.19 g•Fluorine: 34.90 gDetermine the empirical (simplest) formula of the compound.Answer:The simplest formula is K2CuF4.Preview Mode
This document has 16 pages. Sign in to access the full document!