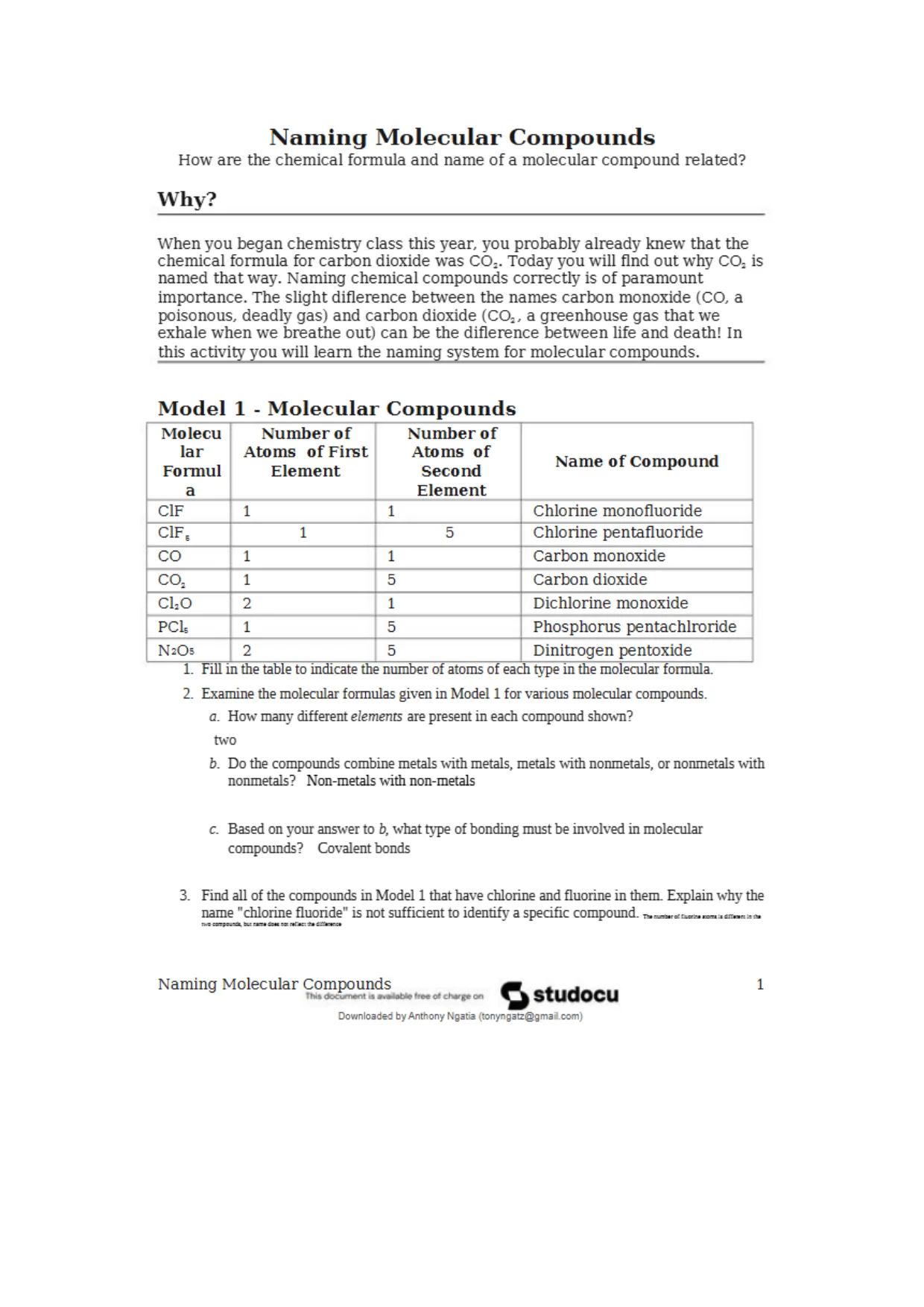
NamingMolecularCompoundsHow are the chemical formula and name of a molecular compound related?Why?When you began chemistry class this year, you probably already knew that thechemical formula for carbon dioxide was COa. Today you will find out why COaisnamed that way. Naming chemical compounds correctly is of paramountimportance. The slight diflerence between the names carbon monoxide (CO, apoisonous, deadly gas) and carbon dioxide (COa, a greenhouse gas that weexhale when we breathe out) can be the diflerence between life and deaths Inthis activity you wTill learn the naming system for molecular compounds.M o d e l 1 - M o l e c u l a r C o m p o u n d sMolecularFormulaNumber ofAtomsof FiretElementNumber ofAtoms ofSecondElementName of CompoundC1F11Chlorine monofluorideC1FE15Chlorine pentafluorideCO11Carbon monoxidecoa15Carbon dioxideCLO21Dichlorine monoxidePCL15Phosphorus pentachlrorideNaOs25Dinitrogen pentoxide1. Fill in the table to indicate the number of atoms of each type in the molecular formula.2. Examine the molecular formulas given in Model 1 for various molecular compounds.a. Haw many different elements are present in each compound shown?twob. Do the compounds combine metals with metals, metals with nonmetals, or nonmetals withnonmetals?Non-metals with non-metalsc.Based on your answer to b, what type of bonding must be involved in molecularcompounds?Covalent bonds3.Find all of the compounds in Model 1 that have chlorine and fluorine in them. Explain why thename "chlorine fluoride1' is not sufficient to identify a specific compound.Mlewrrm!*::±ji12Xa:r*£«Naming Molecular CompoundsriHiLT’writ>il eo i ctwge o nQstudocuDownloaded by Anthony Ngatia {tDnyrgatziggmai.Kjni)
NamingMolecularCompoundsHow are the chemical formula and name of a molecular compound related?Why?When you began chemistry class this year, you probably already knew that thechemical formula for carbon dioxide was COa. Today you will find out why COaisnamed that way. Naming chemical compounds correctly is of paramountimportance. The slight diflerence between the names carbon monoxide (CO, apoisonous, deadly gas) and carbon dioxide (COa, a greenhouse gas that weexhale when we breathe out) can be the diflerence between life and deaths Inthis activity you wTill learn the naming system for molecular compounds.M o d e l 1 - M o l e c u l a r C o m p o u n d sMolecularFormulaNumber ofAtomsof FiretElementNumber ofAtoms ofSecondElementName of CompoundC1F11Chlorine monofluorideC1FE15Chlorine pentafluorideCO11Carbon monoxidecoa15Carbon dioxideCLO21Dichlorine monoxidePCL15Phosphorus pentachlrorideNaOs25Dinitrogen pentoxide1. Fill in the table to indicate the number of atoms of each type in the molecular formula.2. Examine the molecular formulas given in Model 1 for various molecular compounds.a. Haw many different elements are present in each compound shown?twob. Do the compounds combine metals with metals, metals with nonmetals, or nonmetals withnonmetals?Non-metals with non-metalsc.Based on your answer to b, what type of bonding must be involved in molecularcompounds?Covalent bonds3.Find all of the compounds in Model 1 that have chlorine and fluorine in them. Explain why thename "chlorine fluoride1' is not sufficient to identify a specific compound.Mlewrrm!*::±ji12Xa:r*£«Naming Molecular CompoundsriHiLT’writ>il eo i ctwge o nQstudocuDownloaded by Anthony Ngatia {tDnyrgatziggmai.Kjni)Preview Mode
This document has 6 pages. Sign in to access the full document!