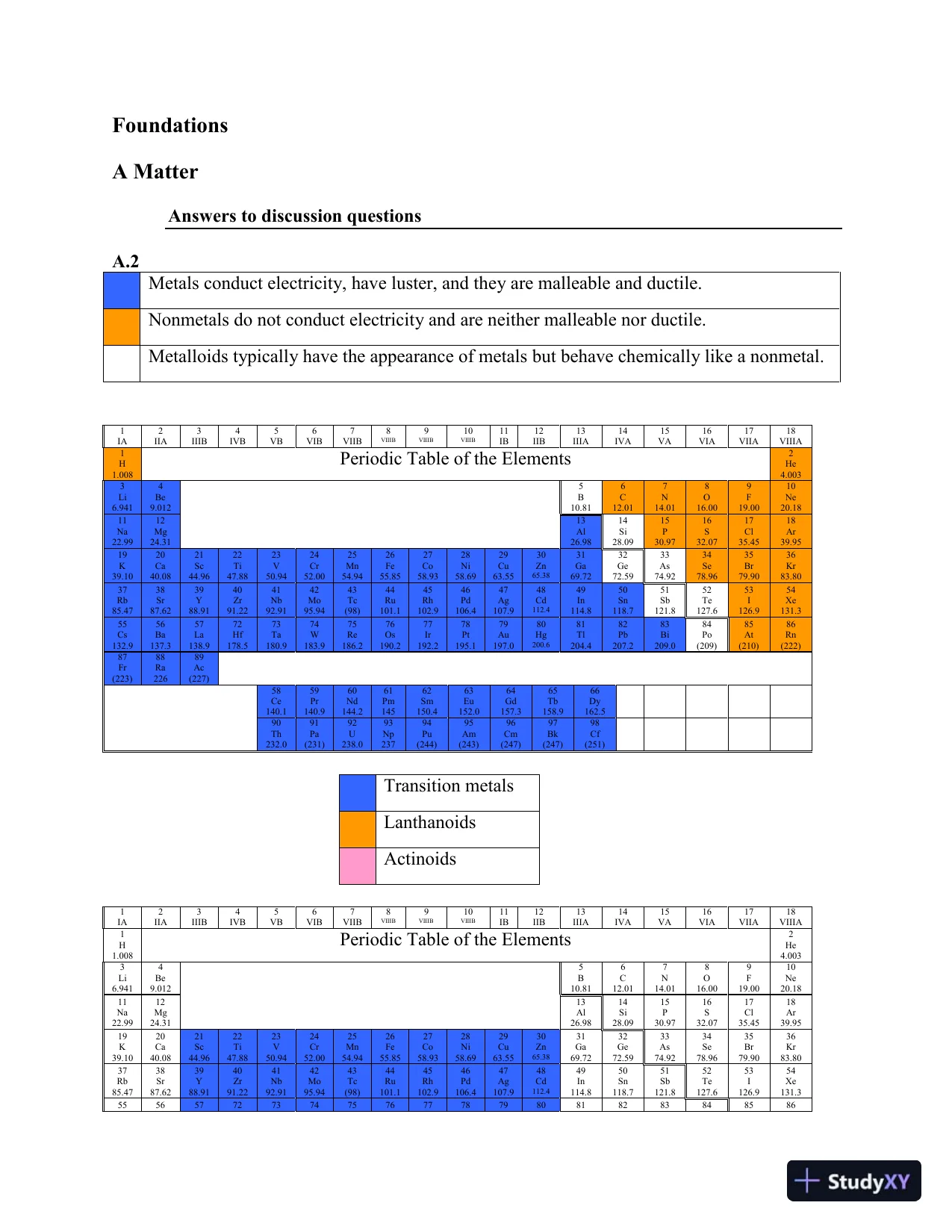
FoundationsA MatterAnswers to discussion questionsA.2Metals conduct electricity, have luster, and they are malleable and ductile.Nonmetals do not conduct electricity and are neither malleable nor ductile.Metalloids typically have the appearance of metals but behave chemically like a nonmetal.1IA2IIA3IIIB4IVB5VB6VIB7VIIB8VIIIB9VIIIB10VIIIB11IB12IIB13IIIA14IVA15VA16VIA17VIIA18VIIIA1H1.008Periodic Table of the Elements2He4.0033Li6.9414Be9.0125B10.816C12.017N14.018O16.009F19.0010Ne20.1811Na22.9912Mg24.3113Al26.9814Si28.0915P30.9716S32.0717Cl35.4518Ar39.9519K39.1020Ca40.0821Sc44.9622Ti47.8823V50.9424Cr52.0025Mn54.9426Fe55.8527Co58.9328Ni58.6929Cu63.5530Zn65.3831Ga69.7232Ge72.5933As74.9234Se78.9635Br79.9036Kr83.8037Rb85.4738Sr87.6239Y88.9140Zr91.2241Nb92.9142Mo95.9443Tc(98)44Ru101.145Rh102.946Pd106.447Ag107.948Cd112.449In114.850Sn118.751Sb121.852Te127.653I126.954Xe131.355Cs132.956Ba137.357La138.972Hf178.573Ta180.974W183.975Re186.276Os190.277Ir192.278Pt195.179Au197.080Hg200.681Tl204.482Pb207.283Bi209.084Po(209)85At(210)86Rn(222)87Fr(223)88Ra22689Ac(227)58Ce140.159Pr140.960Nd144.261Pm14562Sm150.463Eu152.064Gd157.365Tb158.966Dy162.590Th232.091Pa(231)92U238.093Np23794Pu(244)95Am(243)96Cm(247)97Bk(247)98Cf(251)Transition metalsLanthanoidsActinoids1IA2IIA3IIIB4IVB5VB6VIB7VIIB8VIIIB9VIIIB10VIIIB11IB12IIB13IIIA14IVA15VA16VIA17VIIA18VIIIA1H1.008Periodic Table of the Elements2He4.0033Li6.9414Be9.0125B10.816C12.017N14.018O16.009F19.0010Ne20.1811Na22.9912Mg24.3113Al26.9814Si28.0915P30.9716S32.0717Cl35.4518Ar39.9519K39.1020Ca40.0821Sc44.9622Ti47.8823V50.9424Cr52.0025Mn54.9426Fe55.8527Co58.9328Ni58.6929Cu63.5530Zn65.3831Ga69.7232Ge72.5933As74.9234Se78.9635Br79.9036Kr83.8037Rb85.4738Sr87.6239Y88.9140Zr91.2241Nb92.9142Mo95.9443Tc(98)44Ru101.145Rh102.946Pd106.447Ag107.948Cd112.449In114.850Sn118.751Sb121.852Te127.653I126.954Xe131.3555657727374757677787980818283848586
FoundationsA MatterAnswers to discussion questionsA.2Metals conduct electricity, have luster, and they are malleable and ductile.Nonmetals do not conduct electricity and are neither malleable nor ductile.Metalloids typically have the appearance of metals but behave chemically like a nonmetal.1IA2IIA3IIIB4IVB5VB6VIB7VIIB8VIIIB9VIIIB10VIIIB11IB12IIB13IIIA14IVA15VA16VIA17VIIA18VIIIA1H1.008Periodic Table of the Elements2He4.0033Li6.9414Be9.0125B10.816C12.017N14.018O16.009F19.0010Ne20.1811Na22.9912Mg24.3113Al26.9814Si28.0915P30.9716S32.0717Cl35.4518Ar39.9519K39.1020Ca40.0821Sc44.9622Ti47.8823V50.9424Cr52.0025Mn54.9426Fe55.8527Co58.9328Ni58.6929Cu63.5530Zn65.3831Ga69.7232Ge72.5933As74.9234Se78.9635Br79.9036Kr83.8037Rb85.4738Sr87.6239Y88.9140Zr91.2241Nb92.9142Mo95.9443Tc(98)44Ru101.145Rh102.946Pd106.447Ag107.948Cd112.449In114.850Sn118.751Sb121.852Te127.653I126.954Xe131.355Cs132.956Ba137.357La138.972Hf178.573Ta180.974W183.975Re186.276Os190.277Ir192.278Pt195.179Au197.080Hg200.681Tl204.482Pb207.283Bi209.084Po(209)85At(210)86Rn(222)87Fr(223)88Ra22689Ac(227)58Ce140.159Pr140.960Nd144.261Pm14562Sm150.463Eu152.064Gd157.365Tb158.966Dy162.590Th232.091Pa(231)92U238.093Np23794Pu(244)95Am(243)96Cm(247)97Bk(247)98Cf(251)Transition metalsLanthanoidsActinoids1IA2IIA3IIIB4IVB5VB6VIB7VIIB8VIIIB9VIIIB10VIIIB11IB12IIB13IIIA14IVA15VA16VIA17VIIA18VIIIA1H1.008Periodic Table of the Elements2He4.0033Li6.9414Be9.0125B10.816C12.017N14.018O16.009F19.0010Ne20.1811Na22.9912Mg24.3113Al26.9814Si28.0915P30.9716S32.0717Cl35.4518Ar39.9519K39.1020Ca40.0821Sc44.9622Ti47.8823V50.9424Cr52.0025Mn54.9426Fe55.8527Co58.9328Ni58.6929Cu63.5530Zn65.3831Ga69.7232Ge72.5933As74.9234Se78.9635Br79.9036Kr83.8037Rb85.4738Sr87.6239Y88.9140Zr91.2241Nb92.9142Mo95.9443Tc(98)44Ru101.145Rh102.946Pd106.447Ag107.948Cd112.449In114.850Sn118.751Sb121.852Te127.653I126.954Xe131.3555657727374757677787980818283848586Preview Mode
This document has 512 pages. Sign in to access the full document!