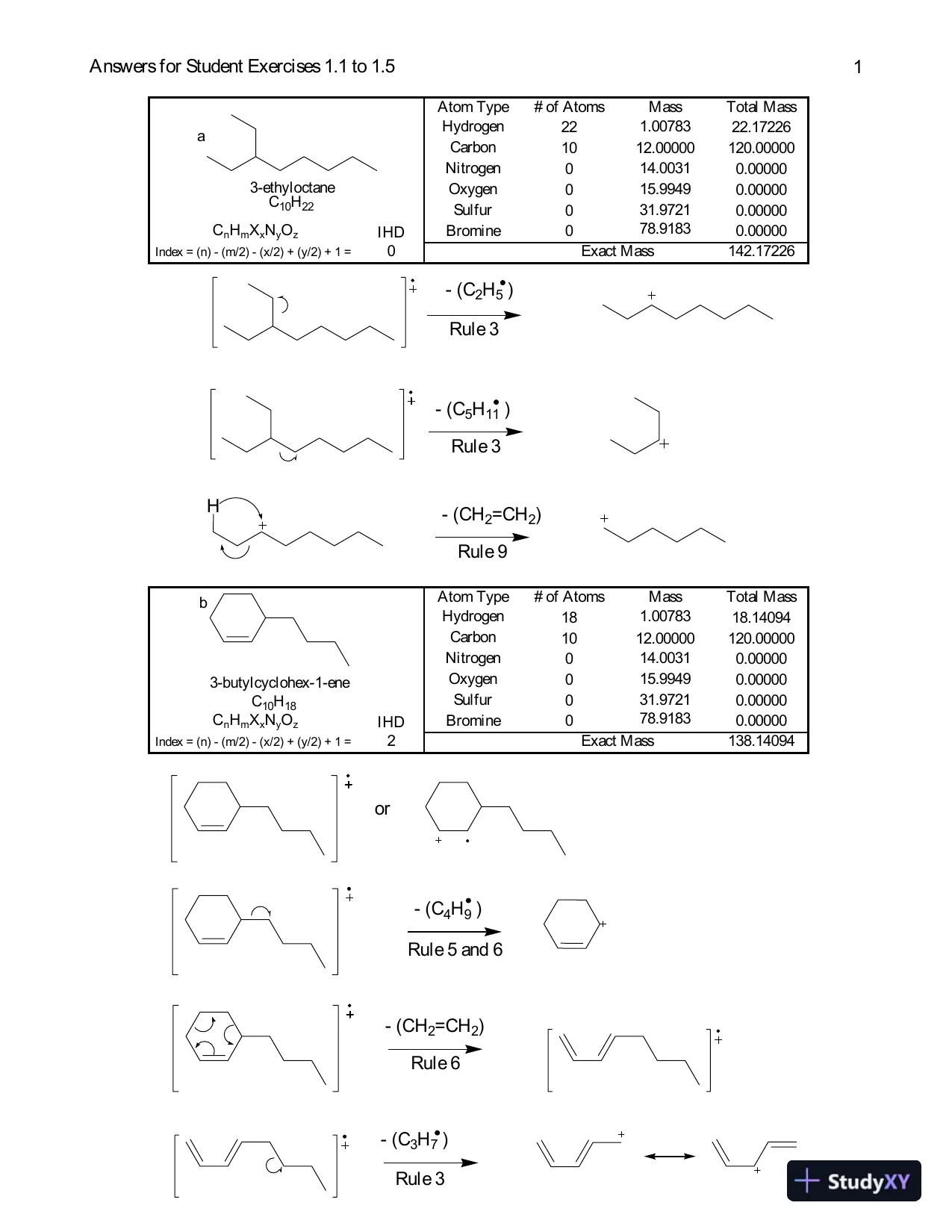
Answers for Student Exercises 1.1 to 1.51Atom Type# of AtomsMassTotal MassHydrogen221.0078322.17226Carbon1012.00000120.00000Nitrogen014.00310.00000Oxygen015.99490.00000Sulfur031.97210.00000CnHmXxNyOzIHDBromine078.91830.00000Index = (n) - (m/2) - (x/2) + (y/2) + 1 =0142.17226Exact Massa3-ethyloctaneC10H22- (C2H5)Rule 3- (C5H11)Rule 3- (CH2=CH2)Rule 9Hor- (C4H9)Rule 5 and 6- (CH2=CH2)Rule 6- (C3H7)Rule 3Atom Type# of AtomsMassTotal MassHydrogen181.0078318.14094Carbon1012.00000120.00000Nitrogen014.00310.00000Oxygen015.99490.00000Sulfur031.97210.00000CnHmXxNyOzIHDBromine078.91830.00000Index = (n) - (m/2) - (x/2) + (y/2) + 1 =2138.14094Exact Massb3-butylcyclohex-1-eneC10H18
Answers for Student Exercises 1.1 to 1.51Atom Type# of AtomsMassTotal MassHydrogen221.0078322.17226Carbon1012.00000120.00000Nitrogen014.00310.00000Oxygen015.99490.00000Sulfur031.97210.00000CnHmXxNyOzIHDBromine078.91830.00000Index = (n) - (m/2) - (x/2) + (y/2) + 1 =0142.17226Exact Massa3-ethyloctaneC10H22- (C2H5)Rule 3- (C5H11)Rule 3- (CH2=CH2)Rule 9Hor- (C4H9)Rule 5 and 6- (CH2=CH2)Rule 6- (C3H7)Rule 3Atom Type# of AtomsMassTotal MassHydrogen181.0078318.14094Carbon1012.00000120.00000Nitrogen014.00310.00000Oxygen015.99490.00000Sulfur031.97210.00000CnHmXxNyOzIHDBromine078.91830.00000Index = (n) - (m/2) - (x/2) + (y/2) + 1 =2138.14094Exact Massb3-butylcyclohex-1-eneC10H18Preview Mode
This document has 242 pages. Sign in to access the full document!