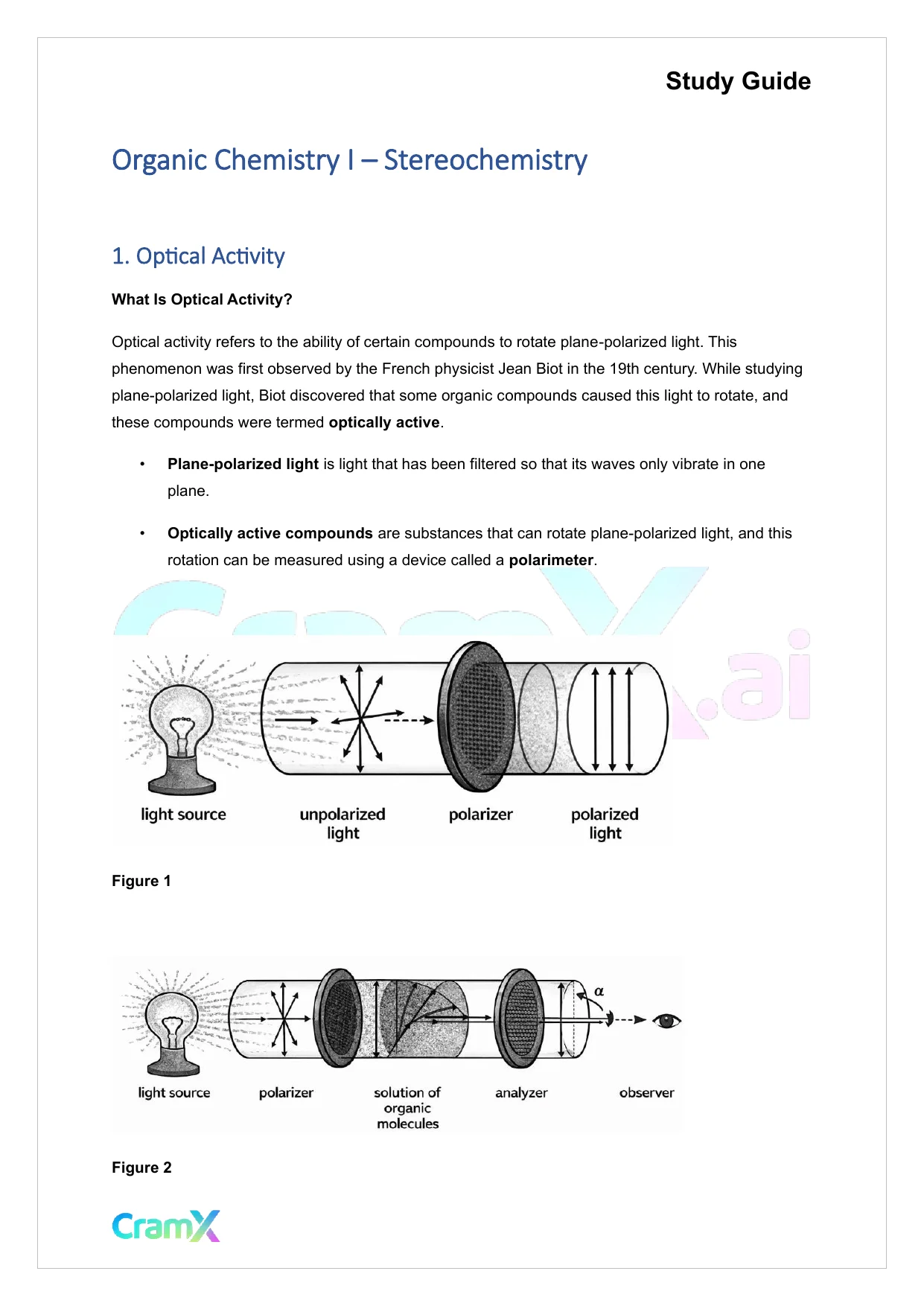
Study GuideOrganic Chemistry I–Stereochemistry1.Optical ActivityWhat Is Optical Activity?Optical activity refers to the ability of certain compounds to rotate plane-polarized light. Thisphenomenon was first observed by the French physicist Jean Biot in the 19th century. While studyingplane-polarized light, Biot discovered that some organic compounds caused this light to rotate, andthese compounds were termedoptically active.•Plane-polarized lightis light that has been filtered so that its waves only vibrate in oneplane.•Optically active compoundsare substances that can rotate plane-polarized light, and thisrotation can be measured using a device called apolarimeter.Figure 1Figure 2
Study GuideOrganic Chemistry I–Stereochemistry1.Optical ActivityWhat Is Optical Activity?Optical activity refers to the ability of certain compounds to rotate plane-polarized light. Thisphenomenon was first observed by the French physicist Jean Biot in the 19th century. While studyingplane-polarized light, Biot discovered that some organic compounds caused this light to rotate, andthese compounds were termedoptically active.•Plane-polarized lightis light that has been filtered so that its waves only vibrate in oneplane.•Optically active compoundsare substances that can rotate plane-polarized light, and thisrotation can be measured using a device called apolarimeter.Figure 1Figure 2Preview Mode
This document has 25 pages. Sign in to access the full document!